On March 31, 2022, Silo Pharma (OTCQB: SILO) announced an agreement with contract research organization Frontage Laboratories for an Investigational New Drug (“IND”)-enabling, “Pharmacokinetic Study.” Silo merges traditional therapeutics with psychedelic research to relieve patients suffering from PTSD, Alzheimer’s, Parkinson’s, and other rare neurological disorders. Frontage will study Silo’s Central Nervous System Peptide, SPU-16, “a potential new treatment for multiple sclerosis and other conditions,” and their Joint Homing Peptide, SPU-21, used for treating arthritogenic processes, which, according to Silo Pharma CEO Eric Weisblum, could, “enhance the therapeutic effect of current and future therapeutics while decreasing potential systemic toxicity….They may be used to treat both Central Nervous System and Autoimmune Diseases.”
Pharmacodynamics studies the reactions between drugs and organisms or, simply, the effect of a drug on the body, allowing for a patient’s existing diseases or conditions, their age and other drugs they’re currently taking. Conversely, pharmacokinetics studies the body’s effect on a drug, its “absorption, distribution, metabolism, and excretion,” or the ways and rates a therapeutic enters, courses through, is modified by and finally exits the system.
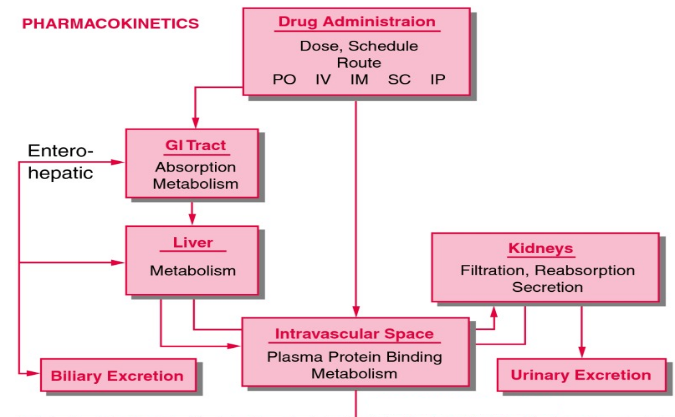
From: Principles of Pharmacokinetics, Copyright © 2003, BC Decker Inc.
DMPK (drug metabolism and pharmacokinetics) constitutes a “core discipline in drug development” for assessing drug safety. According to a Frontage Labs factsheet, DMPK studies provide critical research data, assisting therapeutics innovators like Silo to reach important milestones and “decision making during drug discovery and development.” Weisblum says the agreement (with Frontage), “significantly advances our Central Nervous System Peptide, SPU-16, and our Joint Homing Peptide, SPU-21 technologies closer to the clinic.”
For a closer look at Pharmacokinetics, check out: The National Library of Medicine, Technology Networks: Drug Discovery, the XenoTech Blog and the Frontage Resource Library.
Silo Pharma, headquartered in Englewood Cliffs, New Jersey, identifies assets to license and also funds research to better serve patients and advance the health care industry. Recent achievements include a research agreement with Columbia University developing psychedelic therapeutics for Alzheimer’s disease, DEA approval advancing (with Zylo Therapeutics) Z-Pod technology for delivering time-released Ketamine or Psilocybin, a patent for Silo’s Central Nervous System Homing Peptide, granted by the United States Patent and Trademark Office and a sponsored research agreement with UMB evaluating the pharmacokinetics of dexamethasone delivered to arthritic rats via liposomes.
For Silo Pharma’s most recent news, click here.
Silo Pharma
560 Sylvan Avenue, Suite 3160
Englewood Cliffs, NJ 07632
For more information, visit the company’s website at www.SiloPharma.com.
NOTE TO INVESTORS: The latest news and updates relating to SILO are available in the company’s newsroom at https://ibn.fm/SILO
About BioMedWire
BioMedWire (BMW) is a bio-med news and content distribution company that provides (1) access to a network of wire services via InvestorWire to reach all target markets, industries and demographics in the most effective manner possible, (2) article and editorial syndication to 5,000+ news outlets (3), enhanced press release services to ensure maximum impact, (4) social media distribution via the Investor Brand Network (IBN) to nearly 2 million followers, (5) a full array of corporate communications solutions, and (6) a total news coverage solution with BMW Prime. As a multifaceted organization with an extensive team of contributing journalists and writers, BMW is uniquely positioned to best serve private and public companies that desire to reach a wide audience of investors, consumers, journalists and the general public. By cutting through the overload of information in today’s market, BMW brings its clients unparalleled visibility, recognition and brand awareness. BMW is where news, content and information converge.
To receive SMS text alerts from BioMedWire, text “Biotech” to 888-902-4192 (U.S. Mobile Phones Only)
For more information, please visit https://www.biomedwire.com
Please see full terms of use and disclaimers on the BioMedWire website applicable to all content provided by BMW, wherever published or re-published: http://BMW.fm/Disclaimer
BioMedWire (BMW)
San Francisco, California
www.biomedwire.com
415.949.5050 Office
Editor@BioMedWire.com
BioMedWire is part of the InvestorBrandNetwork.












